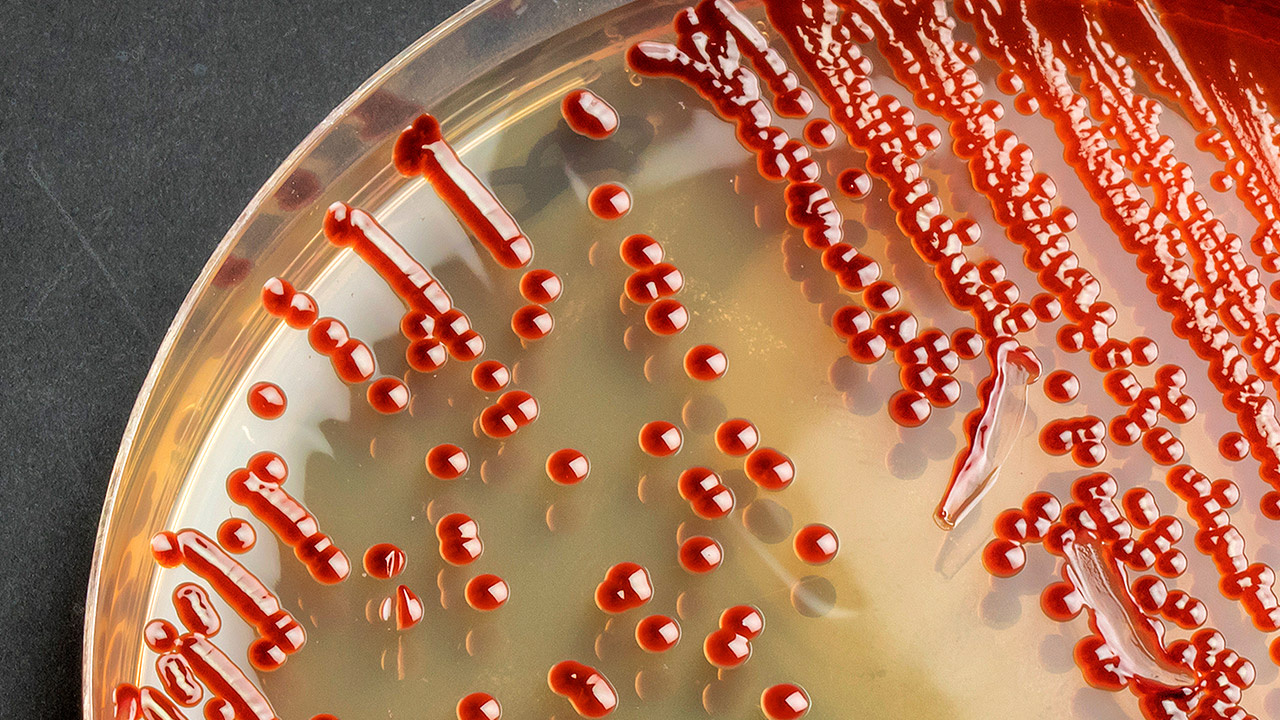
An Environmental Monitoring (EM) program provides meaningful information on the quality of the aseptic processing environment as well as environmental trends of ancillary clean areas. Environmental monitoring should promptly identify potential routes of contamination, allowing for implementation of corrections before product contamination occurs.The establishment of the EM program, training of personnel, data assessment, investigations and product disposition as defined by regulatory organizations highlight course content.
Personnel in manufacturing, Quality control, Quality Assurance, Investigations and CAPA resolution and Regulatory Affairs will gain important information in the establishment and implementation of an environmental monitoring program.
This two day,16 hour course, covers the following topics:
- Importance of microbiological evaluation program for controlled environments
- Critical factors involved in design and implementation of a EM control program
- Review regulatory requirements for environmental monitoring programs
- Establishment of cleanrooms classification and validation requirements
- Sanitization program for EM
- Training and certification of
- personnel (manufacturing and QA/QC)
- Establishment of sampling plan and sites
- Establishment of Alert and Action levels
- Data trending/analysis
- Methodology for quantitative sampling of airborne and surface microorganisms
- Methodology and Instrumentation for quantitative sampling of non-viable total particulates
- Investigations and assignable causes and corrective actions
Upon successful completion, participants will be able to:
- Determine the necessary components for an effective environmental monitoring program for aseptic filling and controlled environments
- Define regulatory expectations of an EM program
- Establish and maintain an environmental monitoring program
- Perform investigations that will withstand regulatory scrutiny ensuring product quality and patient safety
Delivered in-person in a classroom or lab setting.
An Environmental Monitoring (EM) program provides meaningful information on the quality of the aseptic processing environment as well as environmental trends of ancillary clean areas. Environmental monitoring should promptly identify potential routes of contamination, allowing for implementation of corrections before product contamination occurs.The establishment of the EM program, training of personnel, data assessment, investigations and product disposition as defined by regulatory organizations highlight course content.
Personnel in manufacturing, Quality control, Quality Assurance, Investigations and CAPA resolution and Regulatory Affairs will gain important information in the establishment and implementation of an environmental monitoring program.
This two day,16 hour course, covers the following topics:
- Importance of microbiological evaluation program for controlled environments
- Critical factors involved in design and implementation of a EM control program
- Review regulatory requirements for environmental monitoring programs
- Establishment of cleanrooms classification and validation requirements
- Sanitization program for EM
- Training and certification of
- personnel (manufacturing and QA/QC)
- Establishment of sampling plan and sites
- Establishment of Alert and Action levels
- Data trending/analysis
- Methodology for quantitative sampling of airborne and surface microorganisms
- Methodology and Instrumentation for quantitative sampling of non-viable total particulates
- Investigations and assignable causes and corrective actions
Upon successful completion, participants will be able to:
- Determine the necessary components for an effective environmental monitoring program for aseptic filling and controlled environments
- Define regulatory expectations of an EM program
- Establish and maintain an environmental monitoring program
- Perform investigations that will withstand regulatory scrutiny ensuring product quality and patient safety